SSGCID
Seattle Structural Genomics Center for Infectious Disease
Adk
- adenylate kinase (Adk):
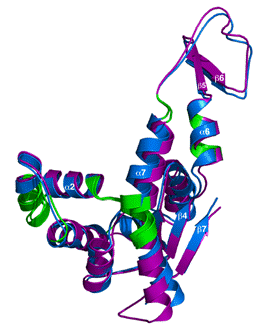
In all organisms adenylate kinases (Adks) play a vital role in cellular energy metabolism and nucleic acid synthesis. Due to differences in catalytic properties between the Adks found in prokaryotes and in the cytoplasm of eukaryotes, there is interest in targeting this enzyme for new drug therapies against infectious bacterial agents. We present here the three-dimensional protein structure of Burkholderia pseudomallei Adk. This pathogen is an aerobic, Gram-negative, soil-dwelling bacterium responsible for the infectious disease melioidosis, which is a serious health problem in Northern Australia and Southeast Asia and accounts for 40% of all sepsis-related mortality in northeast Thailand. Due to its potential use in biological warfare and biological terrorism, B. pseudomallei is also of concern to the U.S. Centers for Disease Control and Prevention. An unexpected feature of the BpAdk crystal structure was the observation of two significantly different conformations of the protein in the same protein crystal. In the figure, the two molecules (purple and blue) are superpositioned. The regions in green are the corresponding highly conserved hinge regions. We propose that the different conformations represent two snapshots of BpAdk as it progresses towards a closed, enzymatically active state. The two conformations of BpAdk observed in the crystal structure reported here represent two more structure snapshots to further study the relationships between enzyme structure, dynamics, and function.
For more information, please see the Protein Data Bank entry 3gmt. This work is published in Biochem Bioph Res Comm (2010) 394:1012–1017.