SSGCID
Seattle Structural Genomics Center for Infectious Disease
RAS GTPase
- RAS-like GTPase Transport Protein:
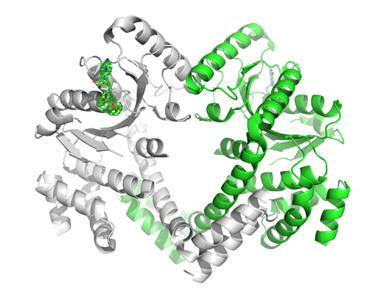
This protein from M. tuberculosis is a member of the Ras super family of proteins. Ras is involved in signal transduction and activating mutations in human Ras are associated with many types of cancer. While originally annotated as an arginine/ornithine transport system ATPase, we show that the protein is most likely a GTPase based on the compound (GDP) bound to the structure. The structure of the GTPase domain of the protein is similar to that of human Ras and several amino acids that line the GTP binding pocket are conserved. Ras-like GTPases use the organic chemical GTP (guanosine-5'-triphosphate) as a regulator of function. During the signal transduction reaction, GTP is converted to GDP (guanosine diphosphate). GDP is shown in the figure bound to the active site of the M. tuberculosisRAS-like GTPase crystal structure. Our structure is related in sequence and structure to the methylmalonic acidemia type A protein (PDB ID 2WWW), a protein which is responsible for a potentially fatal metabolic disorder in humans.For more information, please see the Protein Data Bank entry 3md0. This was also published in 2015:
Crystal structures of Mycobacterial MeaB and MMAA-like GTPases. Edwards, T.E., Baugh, L., Bullen, J., Baydo, R.O., Witte, P., Thompkins, K., Phan, I.Q., Abendroth, J., Clifton, M.C., Sankaran, B., Van Voorhis, W.C., Myler, P.J., Staker, B.L., Grundner, C., Lorimer, D.D. (2015) J Struct Funct Genomics 16: 91-99 (PubMed: 25832174).